Three strategic pathways to navigate Vietnam’s Medical Device Market
Vietnam’s medical device market is booming—expected to reach nearly US$2.5 billion by 2030, with 90–95% of equipment imported. As the MoH tightens regulations under Decree 98/2021 and its amendments, the path to market access is more structured than ever.
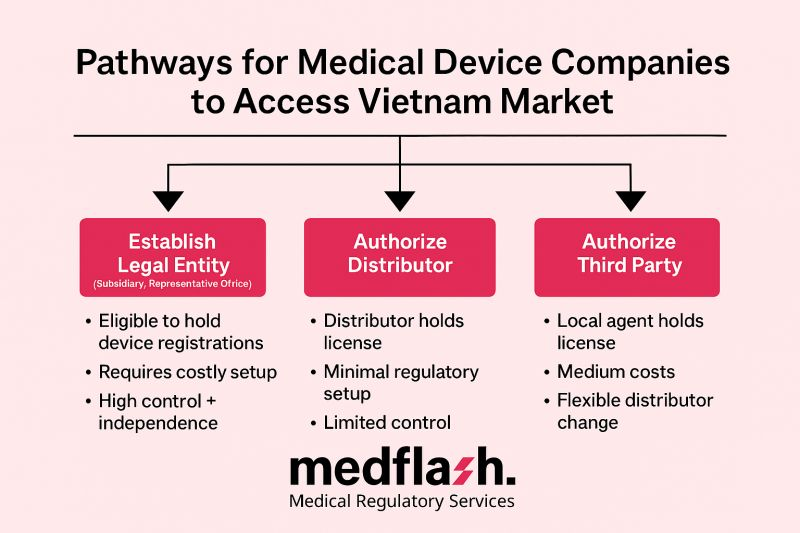
1. Establish a Legal Entity (Subsidiary, Representative Office)
- Eligible to hold device registrations (MA license) + registration holder status.
- Requires local investment certificate and business license.
- High control + independence in pricing, strategy, supply chain—but costly setup, operational burden, QMS localization.
2. Appoint a Local Distributor (Importer)
- Distributor holds import license, manages MA filing and ministry liaison.
- Minimal regulatory setup for foreign company, leverages distributor’s local networks.
- A trade-off: distributor usually owns the MA, potentially reducing your control over pricing and partnerships.
3. Engage an Independent Third-Party Authorized Representative
- Local agent (Medflash) holds marketing authorization in your name, acts as official liaison.
- Switch distributors freely without re-registering products.
- Manages dossier submission, post-market surveillance, renewals, recalls.
- Flexible & cost-effective—ideal for export-phased strategies.
Strategic Considerations
- Large-Tier players: May prefer full control via local entity.
- Export-minded or flexible portfolios: Independent AR keeps you agile and responsive.
- Low-volume/niche launches: Go-to-market with distributor support offers speed.
- Long-term presence: A legal entity signals commitment; greater control over procurement.
Looking to enter Vietnam’s medical device market without the hassle? Let Medflash be your trusted MA holder and regulatory partner.
