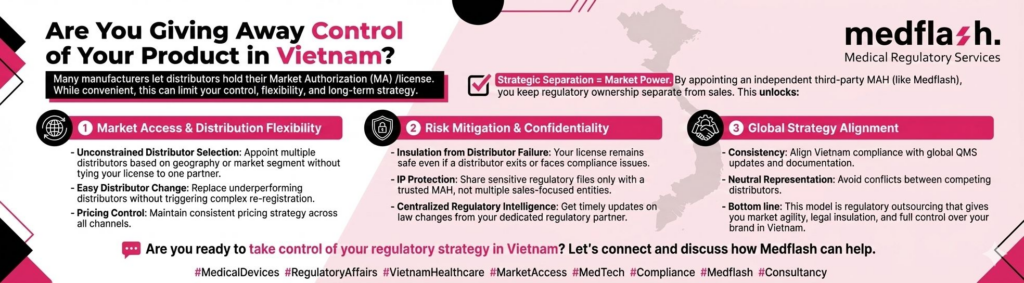
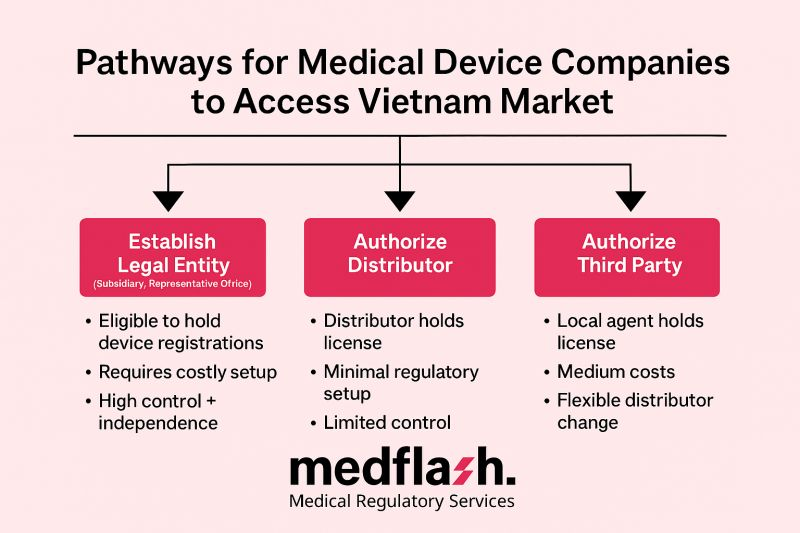
Navigating Borderline Medical Devices: Global Regulatory Insights
Borderline medical devices—products that blur the lines between medical devices, medicinal products, cosmetics, or other categories—pose unique challenges for manufacturers and regulators. Regulatory authorities worldwide provide guidance, manuals, and case studies to clarify classifications, helping ensure compliance and market access. Below, we summarize key resources from major regions and countries, with links to access these critical documents for navigating borderline medical device classifications.
European Union (EU): Comprehensive Classification Manual
The European Commission’s Manual on Borderline and Classification in the Community Regulatory Framework for Medical Devices, published by the Medical Devices Coordination Group (MDCG), is a cornerstone resource. Updated regularly, it includes case studies on products like AED storage units (Class I medical device), lubricants for vaginal dryness (Class IIa), and riboflavin solutions (medicinal product). This manual, aligned with Regulation (EU) 2017/745 (MDR) and Regulation (EU) 2017/746 (IVDR), offers non-binding but consensus-driven guidance.
United Kingdom (UK): MHRA Borderline Determinations
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) provides the “Borderlines with medical devices and other products in Great Britain” document, detailing classifications under the UK Medical Device Regulations 2002. Examples include ethyl chloride sprays (active medical devices) and gym equipment with heart rate monitors (non-medical devices). The MHRA’s Annex A lists specific determinations, offering clarity for manufacturers in Great Britain.
ASEAN Region: Harmonized Classification Efforts
The ASEAN Medical Device Directive (AMDD) guides classification across member states like Singapore and Malaysia. While no centralized list exists, guidance addresses borderline products like hand sanitizers (biocides vs. medical devices) and herbal syrups (food supplements vs. devices). National authorities provide specific insights.
United States (US): FDA Classification Resources
The U.S. Food and Drug Administration (FDA) addresses borderline devices through guidance documents and its Product Classification Database, without a dedicated list. Software as a medical device (SaMD), combination products like drug-eluting stents, and cosmetic lasers are common borderline cases. Manufacturers can request a Request for Designation (RFD) for clarity via the Office of Combination Products (OCP).
Australia: Rules for boundary and combination products
The Therapeutic Goods Administration (TGA) regulates medical devices under the Therapeutic Goods Act 1989, with guidance on borderline products distinguishing devices from medicines or cosmetics.
Brazil: requests for classification of border products
Brazil’s National Health Surveillance Agency (ANVISA) oversees medical devices under resolutions like RDC 751/2022, with borderline products evaluated based on specific procedures. Manufacturers can request for classification of border products.
Canada: Health Canada’s Case-by-Case Approach
Health Canada regulates medical devices under the Medical Devices Regulations, addressing borderline cases through guidance documents like “Classification of products at the drug-medical device interface”, “Classification of Products at the Cosmetic-Drug Interface”. Health Canada evaluates products case-by-case, focusing on intended purpose and risk.
Conclusion
Defining the regulatory status of borderline products requires careful consideration of intended purpose, mechanism of action, and specific definitions within each jurisdiction. Consulting official guidance and classification rules is a vital first step globally.
Navigating these borderline issues can be complex. Don’t hesitate to seek expert regulatory advice to ensure you’re on the right path!